|
30, the surface dilatational viscosity of a asphaltene adsorbed water drop in the oil phase has a typical value of around 0.48 mNs / m. We illustrate this technique using DPPC as the insoluble surfacant monolayer and measured for it a surface dilatational viscosity in the LE phase that is 20 surface poise. In these systems, the dilatational viscosity will be important when the bulk viscosity is relatively small and the surface area to volume ratio of the drop is large. Under these conditions we show that the total tension is uniform along the surface and that the Young-Laplace equation governs the drop shape with the equilibrium surface tension replaced by the constant surface isotropic stress. Compression rates are undertaken slow enough so that bulk hydrodynamic stresses are small compared to the surface tension force. The isotropic total tension on the surface consists of the equilibrium surface tension and the tension due to the dilation. In this presentation we introduce a new technique for measuring the surface dilatational viscosity by contracting an aqueous pendant drop attached to a needle tip and having and insoluble surfactant monolayer at the air-water interface. Surface dilatational viscosity measurements of existing techniques differ by five orders of magnitude and use spatially damped surface waves and rapidly expanding bubbles. On the other hand, surface dilatational viscosity measurements are difficult because a flow which change the surface area also changes the surfactant surface concentration creating changes in the equilibrium interfacial tension that must be also taken into account. The simplest relationship between surface strain rate and surface stress is the Boussinesq-Scriven constitutive equation completely characterized by three coefficients: equilibrium interfacial tension, surface shear viscosity, and surface dilatational viscosity Equilibrium interfacial tension and surface shear viscosity measurements are very well established. When surfactants are subjected to shear and dilatational flows, flow induced interaction of the surfactants can create interfacial stresses apart from the equilibrium surface tension. 1 Viscosity quantifies the internal frictional force between adjacent layers of fluid that are in relative motion. For liquids, it corresponds to the informal concept of 'thickness': for example, syrup has a higher viscosity than water. 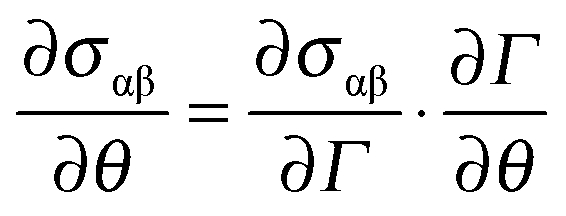
Thus its SI units are newton-seconds per square metre, or pascal-seconds. The viscosity of a fluid is a measure of its resistance to deformation at a given rate. 
Measurement of the Surface Dilatational Viscosity of an Insoluble Surfactant Monolayer at the Air/Water Interface Using a Pendant Drop Apparatus When a fluid interface with surfactants is at rest, the interfacial stress is isotropic (as given by the equilibrium interfacial tension), and is described by the equation of state which relates the surface tension to the surfactant surface concentration. Viscosity is defined scientifically as a force multiplied by a time divided by an area.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
